
Europe has taken a decisive step in the global fight against so-called “forever chemicals”, tightening environmental limits on a class of pollutants that have proven notoriously difficult to remove from water.
The Council of the European Union has formally adopted a directive that expands and updates the list of substances posing risks to surface water and groundwater.
The changes strengthen monitoring, reporting and environmental quality standards across the European Union and bring three cornerstone laws – the Water Framework Directive, the Groundwater Directive and the Environmental Quality Standards Directive – into line with the latest scientific understanding.
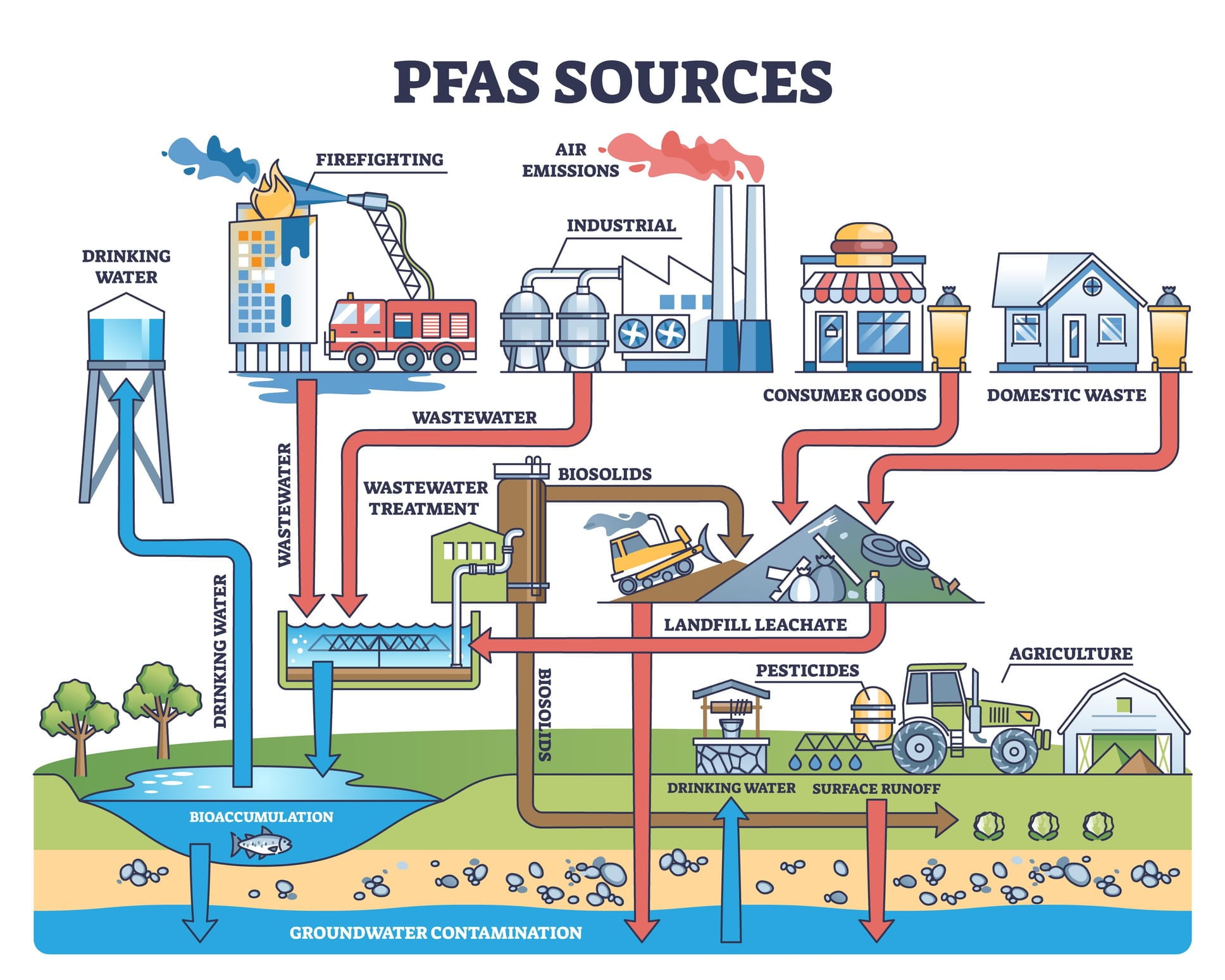
Among the most significant additions are per- and polyfluoroalkyl substances, known as PFAS, a large family of highly persistent chemicals used in products ranging from firefighting foams to industrial coatings.
For water scientists, the decision represents more than a regulatory update. It signals a turning point in how governments approach PFAS pollution, and it may have direct implications for countries such as Australia.
Monash researchers say the directive reflects a growing recognition that PFAS contamination is not simply a localised problem, but a systemic water-quality challenge that demands new treatment technologies and new ways of thinking about environmental protection.
A pollutant that behaves differently
PFAS compounds are unlike most contaminants historically managed in water systems.
They’re highly resistant to chemical breakdown, capable of persisting for decades in rivers, reservoirs and aquifers. They’re also extremely mobile, travelling easily through water and soil. And rather than existing as a single compound, PFAS refers to thousands of chemically-related substances.
Water quality specialist Dr Arash Zamyadi says these properties make PFAS uniquely difficult to manage.
“These compounds are highly persistent, resistant to conventional breakdown processes and often shift between phases rather than being destroyed,” he explains.
“That means we can’t rely on legacy treatment thinking. We need innovative approaches that capture, concentrate and ultimately eliminate them safely.”
The EU directive reflects this scientific reality by requiring regulators to assess the combined risk of chemical mixtures rather than evaluating individual compounds in isolation.
In natural water systems, PFAS rarely occur alone. Instead, many different compounds appear together, and their combined ecological or health effects can be difficult to predict using traditional risk-assessment approaches.
The new regulatory framework acknowledges that complexity.
When monitoring is no longer enough
As environmental thresholds become stricter, simply measuring contamination may not be sufficient to meet regulatory requirements.
Treatment becomes unavoidable.
“Once regulatory limits become sufficiently low, monitoring alone cannot ensure compliance,” says Dr Zamyadi.
“PFAS does not naturally degrade in typical treatment systems. Effective compliance will require integrated solutions that enhance removal efficiency while remaining scalable, energy-conscious and suitable for real wastewater conditions.”
The challenge is particularly acute because conventional treatment methods struggle to remove many of the PFAS compounds now being detected in the environment.
Activated carbon adsorption – widely used in water treatment plants – can capture many long-chain PFAS molecules. But it is significantly less effective against the shorter-chain compounds that increasingly dominate contamination profiles.
That technical limitation means the regulatory shift now underway in Europe is exposing a longstanding engineering challenge.
Rethinking filtration technology
Addressing that challenge is the focus of research led by Professor Mainak Majumder, of Monash Mechanical and Aerospace Engineering, and Director of the Advanced Manufacturing with 2D Materials ARC Research Hub.
His team has been developing a graphene-based nanofiltration membrane designed specifically to capture PFAS molecules from water.
The membrane creates nanoscale channels and chemical binding interactions that block PFAS compounds while allowing clean water to pass through.
“Our FluoroBlock membrane technology simultaneously removes and concentrates both short- and long-chain PFAS contaminants from mixtures,” Professor Majumder explains.
“It can also handle co-contaminants such as natural organic matter and hardening ions like calcium and magnesium in a single-step treatment process.”
Crucially, the technology operates with significantly lower energy requirements than conventional reverse-osmosis systems, making it potentially suitable for a wide range of applications, from large treatment plants to smaller or remote water purification systems.
Read more: Turning dirty water clean: Portable water filter to aid disaster relief efforts
Another key advantage is manufacturability.
Unlike many advanced materials that remain confined to laboratory experiments, the graphene membrane can be produced using scalable printing processes. That opens the possibility of deployment across municipal treatment plants, industrial wastewater facilities and landfill leachate systems.
Monash researchers are currently collaborating with Australian water technology company Clean TeQ Water to manufacture and commercialise the filtration platform.
However, Professor Majumder notes that filtration is only part of the solution.
Captured PFAS must still be destroyed.
“To fully address the problem, removal technologies must ultimately be integrated with degradation processes capable of breaking down these persistent toxic compounds into benign byproducts,” he says.
Why Europe’s policy matters globally
Europe’s new directive is notable not only for what it regulates, but how.
Rather than focusing primarily on drinking water standards, the EU framework emphasises protection of entire water bodies including rivers, lakes and groundwater systems.
That ecosystem-based approach effectively moves regulation upstream.
Instead of managing contamination only at the point where water enters treatment plants, regulators must address PFAS across the broader water cycle – industrial discharge, wastewater treatment, groundwater extraction and recycled water systems.
Compliance therefore becomes an engineering capability challenge as much as a monitoring one.
Dr Zamyadi suggests European environmental standards frequently become global reference points, particularly where environmental compliance intersects with trade, food safety and market access.
“The EU’s precautionary regulatory framework often influences policy beyond its borders,” he says.
“When environmental standards become integrated with international trade and supply chains, other jurisdictions tend to follow.”
In that sense, the directive may foreshadow regulatory changes elsewhere, including Australia.

Australia at a policy crossroads
Australia currently manages PFAS largely through drinking-water guideline values and site-specific contamination responses.
But the European model adopts a broader perspective, recognising PFAS as a persistent contaminant circulating through entire catchment systems.
“A drinking-water lens focuses on the point of human exposure,” says Dr Zamyadi.
“An ecosystem-based framework recognises PFAS as a contaminant that cycles through rivers, sediments, aquifers and even biological systems.”
Managing contamination only at the treatment-plant boundary, he argues, means responding after pollution has already spread.
Protecting entire water bodies shifts the emphasis toward source control, cumulative pollution reduction and long-term environmental resilience.
It may also reduce future financial liabilities.
“Once PFAS accumulates in aquifers or river sediments, remediation becomes technically complex and economically burdensome,” Dr Zamyadi says.
“Managing contamination at the ecosystem scale is both environmentally responsible and a form of risk management.”
Technology and policy moving together
Historically, regulators have often hesitated to impose strict environmental limits where viable treatment technologies were uncertain.
That dynamic may now be changing.
Advances in filtration, oxidation and hybrid treatment systems are beginning to demonstrate that meaningful PFAS removal may be achievable without prohibitive energy costs.
At Monash, researchers are also exploring complementary treatment approaches, including ozone nanobubble and granular activated carbon platforms designed to improve removal efficiency and extend the life of filtration materials.
While promising, these technologies still face challenges, particularly in scaling from laboratory success to full-scale infrastructure.
For Dr Zamyadi, solving PFAS contamination will ultimately depend on collaboration.
“Progress doesn’t rely on scientific discovery alone,” he says. “It requires trusted partnerships between utilities, researchers and technology developers.”
“When industry is willing to pilot, test and refine emerging solutions, complex environmental challenges like PFAS move from being persistent problems toward solvable engineering tasks.”
As Europe pushes ahead with stricter regulation, that partnership between science and policy may become increasingly important not only for Europe, but for water systems around the world.





