
The Andes virus outbreak linked to the expedition cruise ship MV Hondius has infected multiple travellers across several countries, triggering international contact tracing and renewed concern about zoonotic spillover.
Health authorities stress the public risk remains low. But the incident has become a reminder of how quickly an infection that begins in wildlife can move through global travel networks, and why scientists increasingly warn the world is entering an era of more frequent spillover events.
But the MV Hondius hantavirus cluster is not the beginning of another COVID-19. The World Health Organisation has confirmed the virus involved is Andes virus, the only hantavirus known to have limited human-to-human transmission after close, prolonged contact.
Passengers onboard the MV Hondius began disembarking in small groups on Sunday morning, heading for the shore in Tenerife, where they were expected to continue on to charter flights back to their home countries.
— ABC News (@ABC) May 10, 2026
ABC News’ Maggie Rulli reports.https://t.co/HB8syg6GgG pic.twitter.com/dymp19A3Hk
The event is still a warning shot. A virus usually confined to rodents entered an international travel network, caused severe disease, and forced cross-border coordination of diagnostics, isolation and evacuation. That is the anatomy of modern spillover risk.
Hantaviruses are not new. They’re enveloped, negative-sense RNA viruses in the Hantaviridae family. Humans are usually infected by inhaling aerosolised particles from rodent urine, faeces or saliva, especially when contaminated dust is disturbed in enclosed spaces.
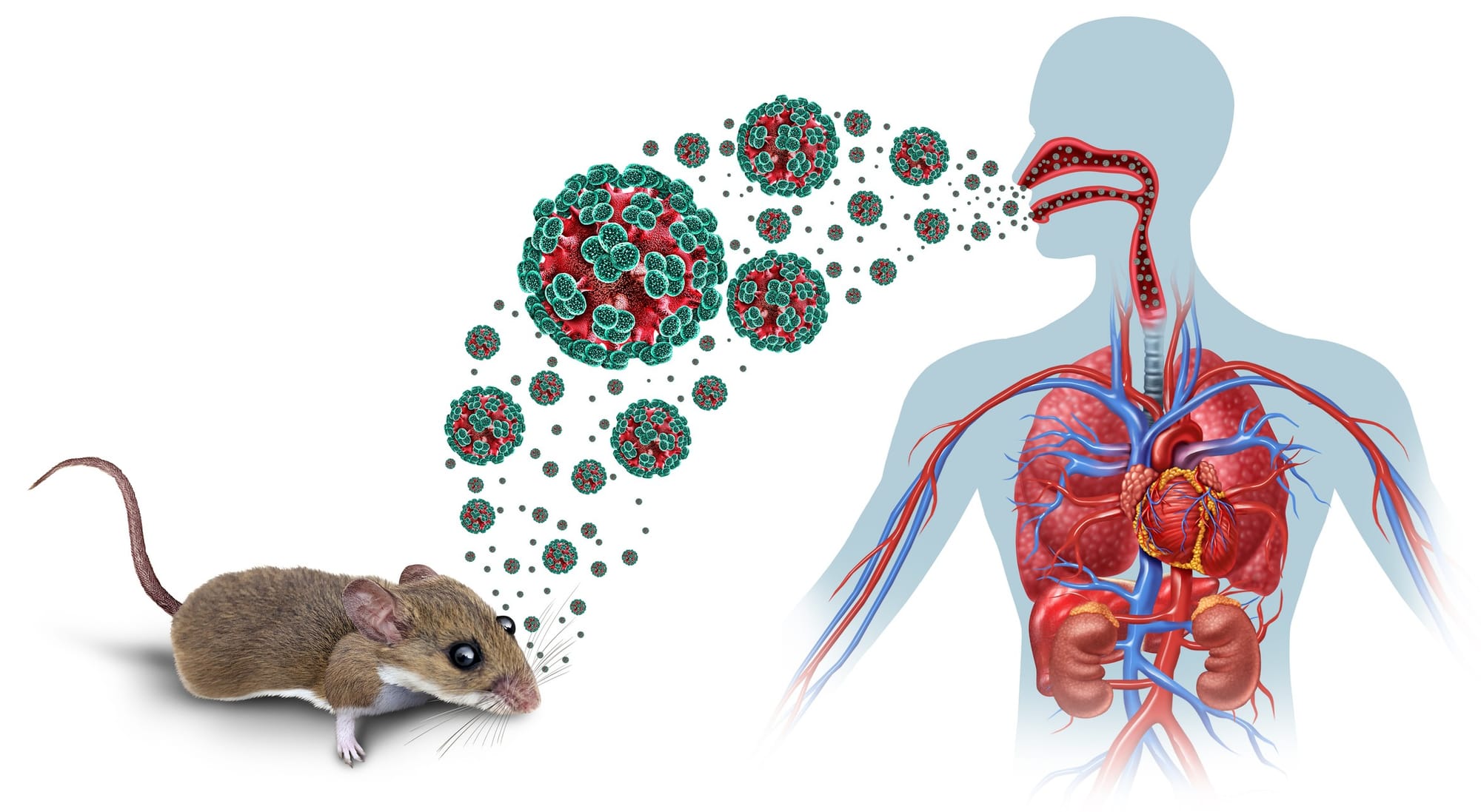
The mechanism is ecological before it is clinical – rodents shed, humans disturb, particles aerosolise, infection follows. Once inside the body, hantaviruses target endothelial cells, causing vascular leak that can lead to renal failure in Europe and Asia or cardiopulmonary collapse in the Americas.
Andes virus changes the risk calculation. Most hantaviruses do not spread efficiently between people, but Andes virus has documented human transmission in Argentina and Chile.
During the 2018-19 Chubut outbreak in Argentina, a single rodent-to-human introduction led to 34 confirmed infections and 11 deaths, amplified by symptomatic people attending crowded social gatherings.
That doesn’t make Andes virus “the next coronavirus”, but it does mean not all hantaviruses can be treated as purely environmental infections. Once a virus gains even limited onward transmission, travel and delayed diagnosis can widen exposure rapidly.
Nach dem Hantavirus-Ausbruch auf der „Hondius“ haben Forscher das Erbgut des Andes-Virus entschlüsselt. Und es gibt neue Verdachtsfälle in Spanien. https://t.co/2biNJ1xg6E pic.twitter.com/0SheavtTMs
— FOCUS online (@focusonline) May 10, 2026
This is no longer an isolated pattern. H5N1 avian influenza has spread from wild birds into poultry, mammals and US dairy cattle, with infectious virus detected in milk and sporadic human infections among exposed workers.
Mpox re-emerged globally in 2022 before triggering a second WHO Public Health Emergency of International Concern in 2024 amid clade I spread in Africa.
Read more: Could monkeypox become the next global pandemic?
Oropouche virus expanded across the Americas, with travel-associated cases and concerns about vertical transmission.
Nipah virus continues to cause lethal outbreaks in Bangladesh and Kerala, while Rwanda’s first Marburg outbreak and Uganda’s Sudan virus outbreak showed how rapidly filoviruses can strain health systems.
Read more: Nipah virus: New cases trigger alert, as scientists uncover how it disarms human defences
The pathogens differ but the signal is the same. Spillover is becoming less a rare accident than a recurring consequence of environmental disruption, intensive farming, global mobility and delayed detection.
The drivers are increasingly clear. Zoonotic emergence is elevated in forested tropical regions undergoing land-use change, where wildlife, livestock and humans are pushed into closer contact.
Climate change is expected to increase cross-species viral sharing as mammals shift ranges and create new ecological encounters.
Long-term Hendra virus data shows how habitat loss and climate stress can push bats closer to horses and people, increasing spillover risk.
Pandemics do not begin only in hospitals; they often begin at forest edges, farms, mines, markets and peri-urban settlements. By the time the first intensive-care patient appears, the ecological event may already be weeks or months old.
This matters because spillover is a biological threshold problem. A virus must encounter a susceptible host, enter the right cells, replicate, evade immunity, shed in sufficient quantity and find a path to the next host. Most viruses fail somewhere along that chain. But repeated exposure gives evolution more chances.
H5N1 in dairy cattle illustrates how an avian virus can establish efficient transmission in a mammalian production system.
Andes virus shows how a rodent-borne pathogen can, under specific conditions, generate human transmission chains. The danger is not that every spillover becomes a pandemic. The danger is that repeated spillovers increase the odds that one pathogen eventually solves enough of the human-transmission puzzle.
For the public, the message should remain balanced. Hantavirus is not spread casually like influenza or SARS-CoV-2. Prevention still depends largely on rodent control, ventilation, disinfectants and protective equipment when cleaning contaminated spaces.
But for public health systems, the lesson is sharper. Preparedness cannot wait for explosive transmission. The critical window is the quiet phase – unexplained fever clusters, dengue-negative illness, pneumonia in travellers, or severe disease among farmers, miners, wildlife workers, veterinarians and healthcare staff.
A pandemic warning does not mean panic. It means building systems that detect early, act locally and share information quickly. That includes One Health surveillance linking wildlife, livestock, environmental and human data; genomic sequencing; rapid diagnostics; occupational protection for high-risk workers; and transparent international reporting.
Pandemic prevention is not only about faster vaccines, but fewer spillovers. Protecting forests, improving farm biosecurity, regulating high-risk wildlife trade and supporting communities at the human-animal interface are public-health interventions, not environmental luxuries.
For Southeast Asia and the broader tropics, the warning is especially relevant. The region sits at the intersection of biodiversity, land-use change, dense mobility, intensive farming and climate-sensitive infections. It’s already experienced SARS, avian influenza, Nipah, dengue expansion and repeated zoonotic alerts.
Malaysia, Singapore and neighbouring countries should view the MV Hondius outbreak not as a distant cruise-ship story, but as a reminder that imported exposure and local diagnostic blind spots can converge quickly. The best defence is not fear, but faster suspicion, better testing and stronger regional coordination.
The next pandemic may not come from hantavirus. It may emerge from influenza, a paramyxovirus, an arenavirus, a coronavirus, an orthopoxvirus, or a virus not yet identified.
But the pattern is visible. Every spillover event is a reminder that ecological disruption has clinical consequences. If outbreaks continue to be treated as isolated surprises, surprise is exactly what will continue to follow.





